 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
 Carbon Steel 4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws
Carbon Steel 4.8/8.8/10.9 Fastener Flower Countersunk Head Self-Tapping Screws
 Carbon Steel Self-Tapping Screws for Kitchen/Cabinet/Furniture
Carbon Steel Self-Tapping Screws for Kitchen/Cabinet/Furniture
 Headless Compression Hollow Screw Cannulated Bone Screws
Headless Compression Hollow Screw Cannulated Bone Screws
 Titanium Cannulated Headless Compression Screw Orthopedic Implant
Titanium Cannulated Headless Compression Screw Orthopedic Implant
 Customizable Steel Black Finish Knurled Thumb Screw with Washer
Customizable Steel Black Finish Knurled Thumb Screw with Washer
 Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
 Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
Customizable Steel Steel Black Finish Knurled Thumb Screw with Washer
 Carbon Steel/4.8/8.8/10.9 M2.5 Galvanized Double Rod Fastener
Carbon Steel/4.8/8.8/10.9 M2.5 Galvanized Double Rod Fastener
The global orthopedic implant market is undergoing a transformative era, with Cancellous Screws playing a pivotal role in trauma and reconstructive surgeries. These specialized fasteners are engineered for fixation in spongy, porous bone structure, distinct from cortical screws used in dense bone. As the world's population ages, the demand for effective treatments for fractures, osteoporosis, and degenerative joint diseases has surged, positioning cancellous screws as a fundamental tool in the modern surgeon's arsenal.
In the commercial sphere, the market is no longer dominated solely by traditional Western medical giants. China has emerged as a powerhouse of orthopedic innovation, blending large-scale manufacturing efficiency with high-end R&D capabilities. This shift has allowed global healthcare providers to access premium-grade implants that meet rigorous international standards like ISO 13485 and CE certifications at a competitive price point.
Cancellous screws find their primary utility in specific anatomical sites where metaphyseal or epiphyseal bone is prevalent:
Procuring medical devices on a global scale requires a deep understanding of quality and logistics. Leading hospitals and distributors prioritize several key factors:
Precision Manufacturing: Tolerance levels in medical screws are measured in microns. Advanced CNC machining is non-negotiable.
Material Traceability: Every screw must be traceable back to its raw material batch to ensure safety and regulatory compliance.
Packaging & Sterility: Ready-to-use, sterile-packed options are increasingly preferred to reduce hospital overhead.
Leveraging decades of industrial expertise, we have redefined what "Made in China" means for the medical sector through a commitment to excellence and technological innovation.
Our "Blue Ocean" strategy focuses on niche orthopedic areas like pediatric trauma and foot surgery. With 33+ patents, we don't just follow trends—we set them.
Equipped with high-precision Swiss-type lathes and cleanroom facilities, we manufacture over 8,000 specifications of orthopedic trauma implants.
From Brazil to Egypt, our export network ensures that critical surgical tools reach hospitals in over 32 countries with localized support.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
The company's self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, and Morocco.

Year Founded

Exp. Countries

Hospitals Served

IP Certificates

Active Agents



 Carbon Steel Galvanized Customized Knurled Bamboo Screw
Carbon Steel Galvanized Customized Knurled Bamboo Screw
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 Hex Socket Head Cap Screw with Grade 12.9 Black
Hex Socket Head Cap Screw with Grade 12.9 Black
 Hex Socket Head Cap Screw with Grade 12.9 Black
Hex Socket Head Cap Screw with Grade 12.9 Black
 Hex Socket Head Cap Screw with Grade 12.9 Black
Hex Socket Head Cap Screw with Grade 12.9 Black
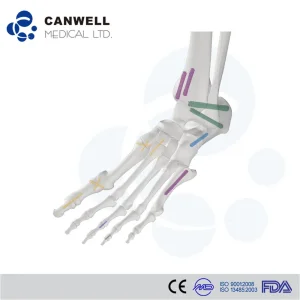 Cannulated Compression Screws, Herbert Screw Headless Screws
Cannulated Compression Screws, Herbert Screw Headless Screws