 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
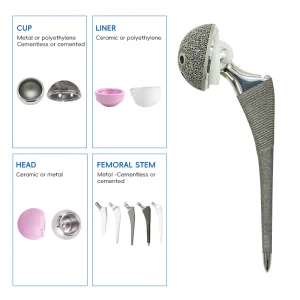 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System German UHMWPE Insert Acetabular Cup Hip Joint System
German UHMWPE Insert Acetabular Cup Hip Joint System Factory Price Tka/Tkr Primary Total Knee Joint Prosthesis
Factory Price Tka/Tkr Primary Total Knee Joint Prosthesis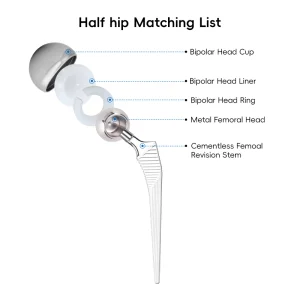 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Set
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Set Orthopedic Total Knee Joint Replacement Instrument Set
Orthopedic Total Knee Joint Replacement Instrument Set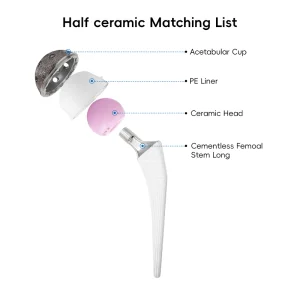 Orthopedic Prosthesis Implant Artificial Hip and Knee
Orthopedic Prosthesis Implant Artificial Hip and Knee Total Hip and Knee Joint Replacement Titanium Implant
Total Hip and Knee Joint Replacement Titanium Implant Manufacturer Rsk Femoral Condyle Revision Tka Replacement
Manufacturer Rsk Femoral Condyle Revision Tka ReplacementUltra-High Molecular Weight Polyethylene (UHMWPE) has revolutionized the medical and industrial sectors over the past five decades. As a premier China UHMWPE liners manufacturer, we recognize that the global demand for high-performance polymers is at an all-time high. In the orthopedic field, UHMWPE liners are the gold standard for bearing surfaces in total hip and knee replacements due to their exceptional wear resistance, biocompatibility, and low friction coefficient.
The global market for UHMWPE liners is expanding rapidly, driven by an aging global population and the increasing prevalence of joint-related ailments such as osteoarthritis. In 2024, the industry is witnessing a shift toward Highly Cross-linked Polyethylene (HXLPE) and Vitamin E-stabilized variations, which significantly reduce oxidative degradation and extend the lifespan of implants. For international procurement officers and medical distributors, sourcing from a specialized factory in China offers a unique balance of advanced material science and cost-efficiency.
The UHMWPE industry is currently navigating several transformative trends. Firstly, the move toward personalized medicine has led to the development of patient-specific UHMWPE liners that accommodate unique anatomical variations. Secondly, the integration of AI-driven manufacturing and CNC precision machining allows for tolerances within microns, ensuring perfect fitment in complex orthopedic assemblies.
Our factory utilizes state-of-the-art electron beam irradiation to achieve optimal cross-linking density. This process enhances the material's structural integrity, making it resistant to the cyclic loading found in human joints. By reducing wear particles, we minimize the risk of osteolysis, which is the primary cause of revision surgeries worldwide.
In global procurement, buyers from the US, Europe, and the Middle East are increasingly looking for "Revision Systems" that utilize high-grade UHMWPE inserts to restore mobility to patients after original implants fail. Our commitment to R&D aligns with these global needs.
Procuring medical-grade UHMWPE liners involves strict regulatory compliance. International buyers require CE and ISO 13485 certifications to ensure patient safety. In regions like Latin America (Brazil, Colombia) and Africa (Egypt, Morocco), there is a growing need for orthopedic implants that are both high-quality and affordable, bridging the gap between top-tier medical care and local economic realities.
Localized application scenarios often vary by anatomy. For instance, the Asian market often requires smaller-sized liners and specific femoral components designed for higher flexion. Our factory excels in OEM/ODM services, allowing us to tailor product specifications—from 8,000 different orthopedic specifications—to meet the localized needs of 31 provinces in China and 32+ countries globally.
As a leading China manufacturer, our strength lies in our integrated supply chain. We don't just produce liners; we manufacture the complete surgical ecosystem, including titanium alloy stems, cobalt-chromium heads, and the precision instruments required for surgery. This vertical integration ensures that every UHMWPE liner is tested in situ within its corresponding assembly.
Our "Focus Strategy" targets the high-precision sectors of pediatric orthopedics and foot/ankle surgery, where UHMWPE must perform under unique mechanical stresses. With 27 national utility model patents, our innovations aren't just theoretical—they are applied in over 300 top-tier teaching hospitals daily.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
The company's self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, and Morocco.

Year Founded

Exp. Countries

Hospitals Served

IP Rights

Global Agents
Quality control in medical-grade UHMWPE production is non-negotiable. Our factory implements a multi-stage verification process. Each batch of UHMWPE resin is tested for molecular weight distribution and density before processing. During machining, we use computerized visual inspection to detect surface micro-cracks or irregularities that could lead to premature wear.
Furthermore, our sterilization processes—using either Ethylene Oxide (EO) or Gamma Irradiation—are conducted in a controlled environment to prevent polymer degradation. This meticulous attention to detail is why we are recognized as one of the best UHMWPE liners manufacturers in China.



 China Factory OEM Support Thr/Tkr Replacement Instrument Set
China Factory OEM Support Thr/Tkr Replacement Instrument Set Orthopaedic Ti-Alloy Cementless Thr Total Hip Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Replacement CE & ISO Certified Hip Joint Prosthesis Replacement Set
CE & ISO Certified Hip Joint Prosthesis Replacement Set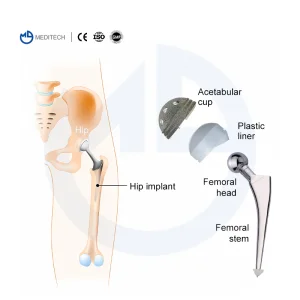 Hip Artificial Knee Joint Orthopedic Cemented Stem Replacement
Hip Artificial Knee Joint Orthopedic Cemented Stem Replacement S-ROM Titanium Alloy Cementless Hip Joints Revision Arthroplasty
S-ROM Titanium Alloy Cementless Hip Joints Revision Arthroplasty Suture Anchor for Shoulder Arthroscopy Implant - Medical Grade
Suture Anchor for Shoulder Arthroscopy Implant - Medical Grade Suture Anchor for Shoulder Arthroscopy Orthopedic Implant
Suture Anchor for Shoulder Arthroscopy Orthopedic Implant Sport Medicine Peek Suture Anchor High Strength Material
Sport Medicine Peek Suture Anchor High Strength Material