 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
 Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
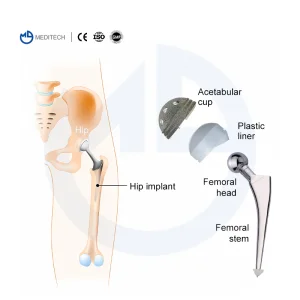 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Artificial Knee Implant Cementless Femoral Stem Knee Replacement Instrument Kit
Artificial Knee Implant Cementless Femoral Stem Knee Replacement Instrument Kit
 CE & ISO Certified Hip Joint Prosthesis Total Hip Replacement Implants
CE & ISO Certified Hip Joint Prosthesis Total Hip Replacement Implants
 Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Implant
Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Implant
 Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
 Veterinary Instrument Orthopaedic Bipolar Hip Prosthesis Replacement
Veterinary Instrument Orthopaedic Bipolar Hip Prosthesis Replacement
The global market for Cementless Hip Stems is currently experiencing an unprecedented era of growth and technological transformation. As a cornerstone of modern Total Hip Arthroplasty (THA), cementless technology—relying on the biological "press-fit" fixation—has become the gold standard for both active younger patients and an aging global population seeking faster recovery times.
In the industrial sphere, the shift from cemented to cementless stems represents a move toward biological longevity. Manufacturers worldwide are investing heavily in material science, specifically focusing on Ti6Al4V Titanium alloys and specialized surface coatings. The commercial demand is driven by a decrease in revision rates and the superior biocompatibility of porous-coated surfaces that encourage direct bone ingrowth (osseointegration).
Key economic drivers include the rising incidence of osteoarthritis and femoral neck fractures globally. Developed markets in North America and Europe maintain a high volume of procedures, while the Asia-Pacific region is emerging as a high-growth manufacturing hub. As an OEM/ODM manufacturer, we bridge the gap between high-end clinical requirements and large-scale industrial production efficiency.
Our flagship Cementless Femoral Stems utilize a dual-layer coating system. The proximal part of the stem is treated with a vacuum plasma-sprayed titanium porous coating followed by a layer of Hydroxyapatite (HA). This combination ensures immediate mechanical stability and long-term biological fixation.
For global distributors and hospital procurement teams, this product offers the perfect balance of geometric design and surface roughness, tailored for various anatomical femoral shapes. It is designed to minimize stress shielding while maximizing proximal load transfer.
The transition from traditional forging to 3D-printed porous structures is the most significant trend. This allows for the creation of trabecular-like surfaces that mimic natural bone architecture, significantly improving the "grip" between the implant and the host bone.
Modern surgery is moving toward patient-specific solutions. Our ODM services focus on producing stems with varying offsets and neck-shaft angles to accommodate diverse global ethnicities and skeletal structures.
Design innovations are now focused on preserving bone stock. Short-stem designs and conservative neck geometries are trending, allowing for easier revision surgery should it be required decades later.
International medical device companies and large-scale hospital groups have evolved in their sourcing strategies. There is a move away from simple "off-the-shelf" purchases toward deep OEM/ODM partnerships. Procurement officers are now looking for manufacturers who can provide:
Our factory in China addresses these needs by combining cost-efficiency with rigorous Swiss-standard quality control, making us a preferred partner for global sourcing hubs.
In complex orthopedic scenarios, such as revision arthroplasty or developmental dysplasia of the hip (DDH), a standard stem is often insufficient. Our S-ROM style modular stems provide surgeons with intraoperative flexibility.
The separate sleeve and stem components allow for independent distal fixation and proximal orientation, ensuring that the patient's unique anatomy is respected. As an OEM factory, we can customize these modular components to fit specific surgical instrument kits and clinical protocols used in different regions.
Application of cementless hip stems varies significantly based on local healthcare infrastructure. In high-income markets, the focus is on "Fast-Track" surgery, where the primary stability of the cementless stem allows patients to bear weight within 24 hours of surgery. This requires high-precision instrumentation that minimizes soft tissue trauma.
In emerging markets, the focus is on the durability and cost-effectiveness of the implant. The "one-and-done" nature of high-quality cementless stems reduces the long-term economic burden of revision surgeries on public health systems. Our products are designed to be intuitive for surgeons, regardless of the technological level of the local surgical theater, supported by our comprehensive instrument sets.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
 2009
Year Founded
2009
Year Founded
 32+
Countries Exported
32+
Countries Exported
 365+
Top-tier Hospitals
365+
Top-tier Hospitals
 30+
IP Certificates
30+
IP Certificates
 300+
Global Agents
300+
Global Agents
Our factory combines the technical prowess of 27 national utility model patent certificates and 6 invention patents with a massive marketing and service network. We don't just manufacture; we educate. Every year, we participate in or host more than 50 academic conferences, ensuring our products are used at the forefront of medical science.
Our self-operated export business serves critical markets in Brazil, Colombia, Peru, Egypt, and Morocco, proving our ability to meet diverse regulatory and logistical challenges.
The manufacturing of a cementless hip stem requires extreme precision. We utilize multi-axis CNC machining centers to ensure the neck geometry and taper dimensions are perfect down to the micron. This is critical for the "morse taper" connection between the stem and the femoral head, preventing fretting corrosion and ensuring mechanical integrity.
Every implant undergoes rigorous inspection, including:

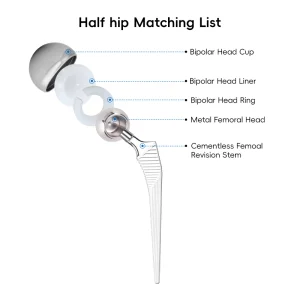 Orthopedic Tkr/Thr Surgery Hip and Knee Instrument Set
Orthopedic Tkr/Thr Surgery Hip and Knee Instrument Set
 Surgery Hip and Knee Prosthesis Instrument Set
Surgery Hip and Knee Prosthesis Instrument Set
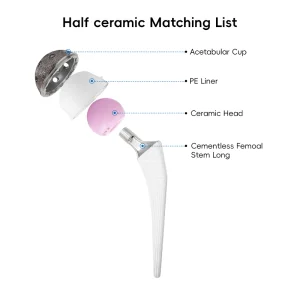 Orthopedic Prosthesis Implant Artificial Joint Replacement
Orthopedic Prosthesis Implant Artificial Joint Replacement
 Total Hip and Knee Joint Titanium Implant China
Total Hip and Knee Joint Titanium Implant China
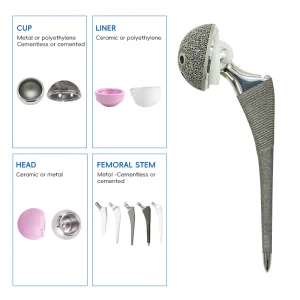 UHMWPE Insert Acetabular Cup Hip Hemiarthroplasty
UHMWPE Insert Acetabular Cup Hip Hemiarthroplasty
 German UHMWPE Insert Acetabular Cup Hip Joint Set
German UHMWPE Insert Acetabular Cup Hip Joint Set
 Instruments for Knee Replacement Surgery Hip System
Instruments for Knee Replacement Surgery Hip System
 China Factory OEM Thr/Tkr Replacement Instrument Set
China Factory OEM Thr/Tkr Replacement Instrument Set
Choosing the right cementless hip stem manufacturer is not just a business decision; it is a clinical commitment to patient health. We stand at the intersection of technological innovation and manufacturing reliability. Our team of over 40 marketing professionals and dedicated technical service personnel in over 20 major cities are ready to support your orthopedic projects.
Whether you require custom ODM designs for specific skeletal morphologies or large-scale OEM production under your own brand, our facility provides the quality, compliance, and capacity needed to succeed in the competitive global orthopedic market. Let us help you bring world-class hip replacement solutions to surgeons and patients worldwide.