 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants

Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner

Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner
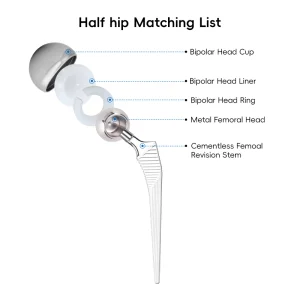
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set

Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set




Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set for Knee Reconstruction
The global orthopedic implant market is witnessing a significant shift toward bone-conserving and ligament-preserving surgical options. Among these, the Cruciate Retaining (CR) Knee system has emerged as a gold standard for patients with a functional Posterior Cruciate Ligament (PCL). Unlike Posterior Stabilized (PS) designs, CR knees allow for more natural kinematics, better proprioception, and reduced bone resection, making them highly sought after in advanced medical markets across North America, Europe, and Asia-Pacific.
Current industrial data suggests that the demand for customized OEM Cruciate Retaining solutions is growing at a CAGR of 6.2%. This surge is driven by an aging global population and an increasing number of younger, active patients requiring knee replacements that can withstand high-demand lifestyles. Manufacturers are now focusing on enhancing wear resistance through advanced cross-linked polyethylene and biocompatible titanium alloys.

Our flagship Cruciate Retaining systems are designed for surgical efficiency. By integrating high-grade materials and ergonomic design, we ensure that the PCL's natural function is maintained, providing patients with a faster recovery time and a more "natural" feel of the joint post-surgery.
As a leading OEM/ODM manufacturer, we offer full customization on femoral component sizing and tibial insert thicknesses to accommodate diverse global patient populations.
The industry is moving toward Robotic-Arm Assisted Surgery. As an ODM supplier, we are optimizing our CR knee designs to be compatible with major navigation and robotic platforms for sub-millimeter precision.
Additive manufacturing (3D printing) allows for porous surfaces that promote osseointegration. Our R&D department is currently integrating 3D-printed titanium structures into our CR tibial trays.
Personalized medicine is no longer a luxury. We provide PSI (Patient-Specific Instrument) services, creating 3D-molds based on CT/MRI data for individualized CR knee fits.

Global procurement teams from top-tier teaching hospitals and medical distributors look for three pillars: Quality Stability, Supply Chain Security, and Cost Efficiency. Our Cruciate Retaining knees are widely used in:
In local application scenarios, the CR design is preferred for its smaller incision requirement and less invasive nature, aligning with the "Day Surgery" trend in modern healthcare.
Choosing us as your China-based OEM/ODM partner grants you access to an unparalleled manufacturing ecosystem. Our factory integrates the entire lifecycle of orthopedic production—from raw material sourcing of medical-grade titanium to 5-axis CNC machining and Class 100,000 cleanroom packaging.
We leverage "Blue Ocean" differentiation strategies, focusing on limb orthopedics and pediatric-specific CR solutions that many Western manufacturers overlook. Our competitive edge lies in our ability to produce over 8,000 product specifications while maintaining rigorous international quality control standards.
Year Founded
Export Countries
Top-tier Hospitals Served
IP Rights & Patents
Global Agents
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
The company's self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, and Morocco.


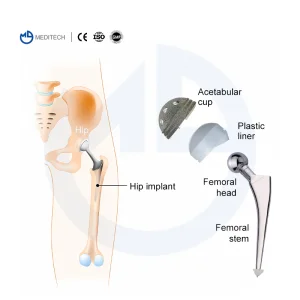
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement

Artificial Knee Implant Cementless Femoral Stem Knee Replacement Instrument Kit


Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka

Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement

Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit

Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit
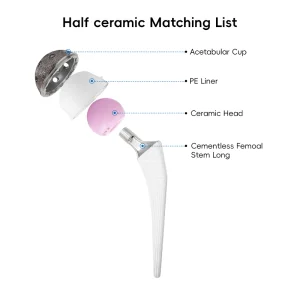
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement