 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants

Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner

Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner
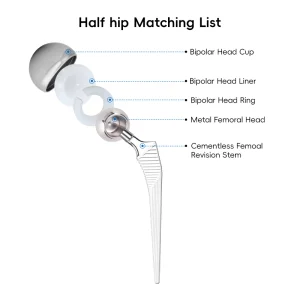
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set

Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set

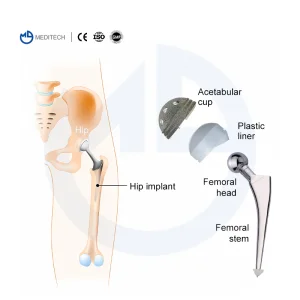
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement


Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set for Knee Reconstruction
The global Total Knee Arthroplasty (TKA) market is witnessing an unprecedented surge, driven by an aging global population and the increasing prevalence of osteoarthritis. As clinical outcomes for knee replacements continue to improve, the demand for high-quality, durable, and biocompatible Total Knee Systems has transitioned from a specialized medical need to a significant global industrial sector. Today, the market is not just about the implants themselves but involves a complex ecosystem of precision instrumentation, specialized positioning tools, and advanced manufacturing technologies.
In developed markets like North America and Europe, the shift is toward outpatient surgeries and rapid recovery protocols. This has increased the demand for highly efficient surgical instrument sets that reduce operative time and improve accuracy. Meanwhile, in emerging economies across Asia, Latin America, and Africa, the focus is on balancing cost-effectiveness with clinical reliability. As a leading OEM/ODM Total Knee System Factory, we bridge this gap by providing high-performance implants and instruments that meet international standards (CE, ISO) while maintaining competitive manufacturing costs.
Accuracy in total knee replacement begins before the first incision is made. Proper limb positioning is critical for surgical access and alignment. Our specialized positioning systems are designed to provide stable, adjustable support for the patient's leg, ensuring the surgical team has an optimal field of view. By integrating these tools into your product portfolio, distributors can offer a complete surgical solution rather than just an implant.
Our commitment to Blue Ocean Strategy ensures we develop niche products that solve real-world surgical challenges, such as the Medical Well-Leg Positioner, which has become a staple in modern orthopedic operating theaters.
The orthopedic industry is currently undergoing a "Digital Revolution." Several key trends are shaping the future of Total Knee Systems:
At our facility, we integrate these trends into our ODM services, allowing our global partners to brand products that are at the absolute cutting edge of medical technology.
Global medical device distributors and hospital procurement departments are under constant pressure to optimize supply chains. They require partners who can offer:
1. Strict Quality Control: Adherence to international medical standards is non-negotiable.
2. Scalable Production: The ability to handle high-volume orders without compromising on lead times.
3. R&D Flexibility: A factory that can customize designs based on local surgical preferences or anatomical variations in different ethnic populations.
Why China? China has become the global hub for orthopedic manufacturing. As a premier China-based factory, we offer a unique synergy of advanced manufacturing infrastructure and cost-efficient production. Our capability covers more than 8,000 product specifications, from trauma and spinal implants to complex total knee replacement systems.
The femoral component is the heart of any TKA system. Our RSK Femoral Condyle Revision system is engineered for maximum stability and physiological movement. Using high-precision CNC machining and vacuum plasma spraying technology, we ensure a surface finish that promotes biological fixation and long-term durability.
Whether you are looking for primary knee systems or revision solutions, our OEM capability allows for specific modifications to the condylar geometry to suit your market's needs.

Year Founded

Export Countries

Top-tier Hospitals

IP Certificates

Global Agents
Total Knee Systems are applied differently across the globe. In Southeast Asia, for example, there is a higher requirement for "High-Flex" knee designs due to cultural practices involving deep squatting and floor-sitting. In contrast, European markets might prioritize implants optimized for high-activity lifestyles and sports.
Our ODM services excel in localized customization. We work with orthopedic surgeons from Brazil to Egypt to understand the nuances of their patient populations. By adjusting the "Blue Ocean" parameters of our product designs, we provide tools and implants that are not just generic medical devices, but specific solutions tailored to local surgical techniques and patient biometrics.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery.
The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights. With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
The company's self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, and Morocco.



As we look forward, the integration of AI-driven diagnostic tools and smart implants is on the horizon. Our R&D department is already exploring sensors that can monitor post-operative joint stability. Partnering with us as your primary Total Knee System Exporter means you are not just buying today's technology, but you are aligning with a future-proof manufacturing powerhouse.
Our comprehensive instrument sets for TKR and THR surgery are designed for durability and ease of use, ensuring that surgeons can perform with confidence. From Titanium knee replacements to UHMWPE acetabular cups, every product that leaves our factory is a testament to our precision-first philosophy.
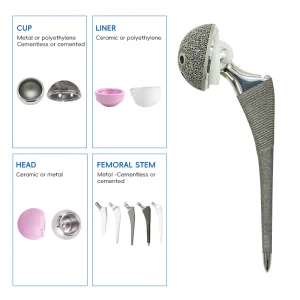

Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China

Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement

Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka

Artificial Knee Implant Cementless Femoral Stem Knee Replacement Instrument Kit

China Factory OEM Support Thr/Tkr Total Knee and Hip Joint Replacement Instrument Set
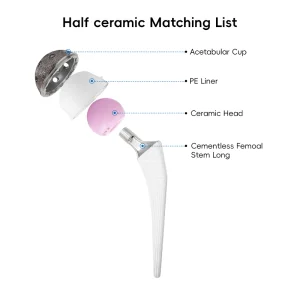
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement

Acl Pcl Reconstruction Instrument Set for Knee Surgery-Complete Orthopedic Ligament Repair Kit