 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants


Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
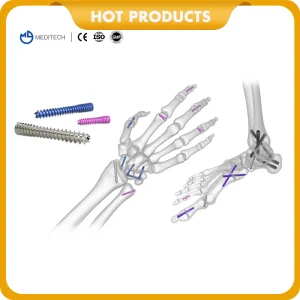
Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
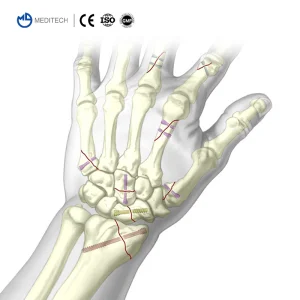
Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price



High Quality 6.5mm Cannulated Screw Instrument Set Orthopedic Surgical Nail Type for Surgery

Orthopedic Locking Screws Surgical Titanium Implant Cannulated Screws for Bone Fracture
In the modern era of orthopedic surgery, cannulated drills and screws represent the pinnacle of minimally invasive internal fixation. These specialized instruments, characterized by their hollow central channel, allow surgeons to insert guide wires first, ensuring pinpoint accuracy before the final implant or drilling occurs. The global market for these devices is experiencing an unprecedented surge, driven by an aging global population and the rising incidence of sports-related injuries.
From North America to the emerging markets of Asia-Pacific, the demand for precision-engineered orthopedic tools is shifting. No longer are generic instruments sufficient; surgeons now demand ergonomic designs, high-torque capabilities, and biocompatible materials like Grade 5 Titanium (Ti6Al4V) and high-grade surgical stainless steel. As a premier manufacturer, we stay at the forefront of this industrial evolution, integrating AI-driven design and robotic manufacturing to produce instruments that meet the stringent requirements of the world's "Top 10" healthcare providers.
The orthopedic industry is witnessing several key trends that are redefining how cannulated drills are manufactured and utilized:
There is a massive push towards smaller incisions. Cannulated systems are the backbone of MIS, allowing for percutaneous fixation which reduces patient recovery time and post-operative pain.
Integration of digital sensors within surgical power tools to monitor bone density and drilling depth in real-time, preventing over-penetration and improving surgical safety.
While standard sizes remain essential, the rise of patient-specific implants is driving the need for versatile cannulated tools that can adapt to unique anatomical structures.
Leading the industry since 2009 with certified quality and worldwide reach.

Year Founded

Export Countries

Top-tier Hospitals

IP Certificates

Global Agents
Procurement officers in international hospitals and medical distribution firms prioritize three pillars: Regulatory Compliance (CE/ISO), Precision Performance, and Supply Chain Reliability. Whether it's a trauma center in Brazil or a pediatric orthopedic clinic in Egypt, the requirements for cannulated drills often center around their versatility in various surgical scenarios.
Common Local Application Scenarios:
As a leading Chinese manufacturer, we offer a "Blue Ocean" strategy that combines innovation with cost-efficiency. Our facility places great importance on scientific and technological innovation, as well as new product research and development. With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, we provide a one-stop-shop for global distributors.
Our strategy follows the principles of "differentiation and focus," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal/external fixation products for foot and ankle surgery. We have obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights, ensuring that our products are not just copies, but patented innovations.

The company's self-operated export business is rapidly expanding. Our sales network already extends to Brazil, Colombia, Peru, Egypt, and Morocco. Our marketing team consists of more than 40 professionals, with technical service personnel present in over 20 major cities, collaborating with more than 300 agents globally.

Quality control is the heartbeat of our factory. Every Cannulated Bone Screw and drill bit undergoes rigorous testing, including fatigue testing, torque resistance analysis, and surface finish inspection. We utilize advanced CNC Swiss-style machining to ensure the concentricity of the cannulated hole—a critical factor for smooth guide wire transition.
Our commitment to education is equally strong. We participate in or host more than 50 academic conferences for orthopedic doctors annually. This feedback loop allows us to refine our instruments based on real-world surgical challenges, ensuring that our "Jin Medical" and "Nx Medical" brands remain synonymous with reliability.

For international buyers looking for a "Top 10" level partner, we offer comprehensive OEM and ODM services. From initial CAD design to sterile packaging and global logistics, our team ensures that every specification—whether it's a 4.5mm cannulated screw or a complex spinal fixation system—is delivered with perfection.

Canwell 4.5mm Orthopedic Plates and Screws 7.3mm Cannulated Screw with Short Thread Instrument Set

Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
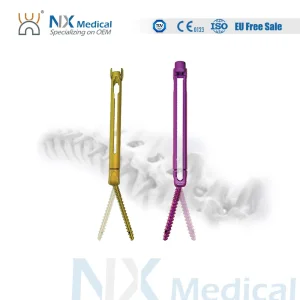
Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw Spinal Fixation

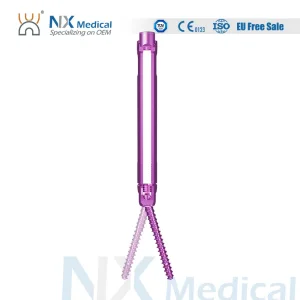
Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
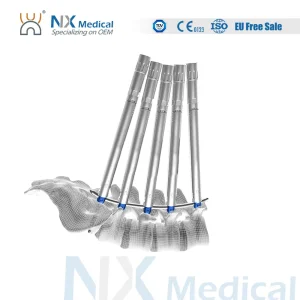
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Orthopedic Internal Fixation

Nx Medical Sf-IV Mis Cannulated Bone Cement Spinal Pedicle Screw 5.5 for Bone Fixation
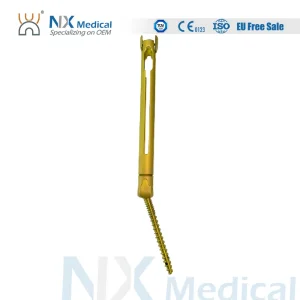
Nx Medical Orthopedic Implant Titanium Cannulated Bone Cement Spinal Pedicle Screw