Deep Dive into Acetabular Shell Engineering & Market Evolution
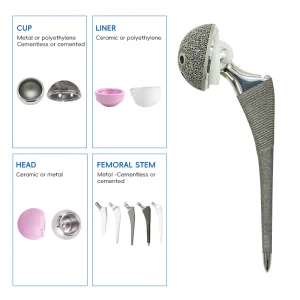
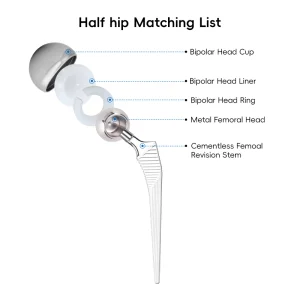
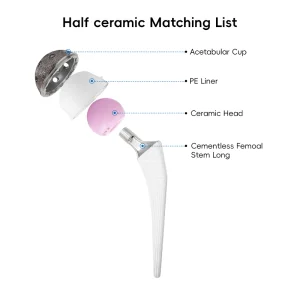
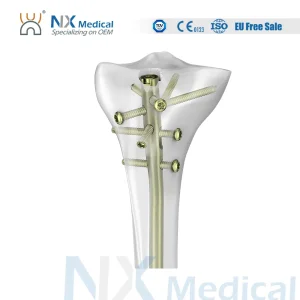
The manufacturing of acetabular shells has undergone a radical transformation over the last decade. As a premier China Acetabular Shells Manufacturer, we have observed the shift from simple cemented components to complex, biologically active surfaces. The modern acetabular shell is not just a metal cup; it is a sophisticated interface between human biology and titanium engineering. Our factory utilizes high-grade medical titanium alloys (Ti6Al4V) which offer the perfect balance of fatigue strength and modulus of elasticity, closely mimicking human bone properties. This reduces the effect of "stress shielding," a common cause of bone resorption around implants.
Our research and development team, backed by numerous national patents, focuses on the micro-topography of the shell's exterior. By applying advanced titanium coating technologies, we create a surface with controlled porosity. This allows the host bone to grow into the implant, creating a permanent, biological bond. This "biologic fixation" is essential for the long-term success of the Total Hip Arthroplasty Artificial Hip Titanium Coating Prosthetics, especially in younger, more active patients who expect their implants to last 20 to 30 years.
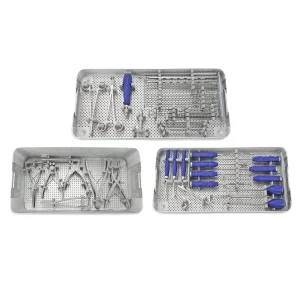
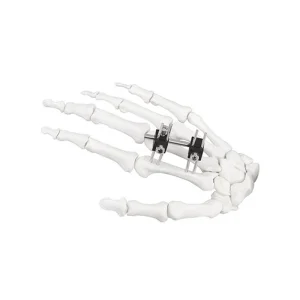
Furthermore, the global procurement landscape is shifting towards integrated systems. Buyers no longer look for just the shell; they require a comprehensive ecosystem including the UHMWPE Insert Acetabular Cup, the femoral stem, and the precise surgical instrument sets required for implantation. Our factory excels in providing this "one-stop" solution. By controlling the entire manufacturing process—from raw material sourcing to the final sterile packaging—we reduce the logistics burden for our international partners. This vertical integration is a key advantage of the Chinese manufacturing model, allowing for rapid scaling and customization that European or American counterparts often struggle to match at the same price point.
In terms of localized application, our products are increasingly used in emerging medical fields. For instance, the Veterinary Surgical Hip Joint Total Replacement market is expanding. Large canine patients suffer from hip dysplasia similarly to humans, and our orthopedic implants have been adapted to meet the anatomical requirements of veterinary medicine. This diversification into veterinary trauma and spinal fixation showcases our flexibility and engineering prowess. Whether it is a Cervical Spine Fixation device or a Proximal Tibial Interlocking Intramedullary Nail, our quality remains consistent, ensuring patient safety across all biological species.
Finally, the importance of academic promotion cannot be overstated. We don't just sell products; we sell clinical success. By participating in over 50 academic conferences annually, we stay connected with the frontline surgeons. This feedback loop allows us to iterate our designs, making them more ergonomic for the surgeon and more effective for the patient. Our export business, reaching from South America to North Africa, is a testament to the global trust in our "Made in China" orthopedic solutions. We continue to invest in our marketing network, serving over 300 top-tier teaching hospitals and ensuring that our technical service personnel are always available to provide support.
 Alur Orthopedic Implants
Alur Orthopedic Implants