 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
The global orthopedic trauma fixation market has witnessed a paradigm shift toward minimally invasive procedures, where Intramedullary Nails (IM Nails) stand as the gold standard for long bone fracture stabilization. As a premier Intramedullary Nails Supplier & Exporter, we recognize that the industry is no longer just about manufacturing hardware; it is about biological integration and biomechanical stability. From the femoral shaft to the distal tibia, IM nails provide the internal splinting required for early weight-bearing and accelerated patient recovery.
Current industrial trends indicate a surge in the adoption of Titanium Alloy (Ti6Al4V) and carbon-fiber reinforced materials. These materials offer the perfect balance of modulus of elasticity (closer to human bone) and fatigue resistance. Furthermore, the integration of AI-assisted preoperative planning and 3D-printed patient-specific guides is revolutionizing how surgeons approach complex comminuted fractures.


Year Founded

Exp. Countries

Top-tier Hospitals

IPR Certificates

Global Agents
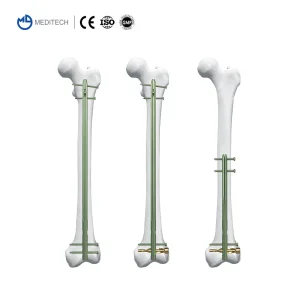
Procurement departments in modern healthcare facilities are increasingly focused on Supply Chain Resilience and Cost-to-Value ratios. For many hospitals in Brazil, Egypt, and Colombia, the challenge lies in obtaining high-quality implants that meet CE and ISO standards without the prohibitive markups of traditional "Big MedTech" brands. This is where our role as a high-capacity Chinese factory becomes vital.
Our Intramedullary Nails are designed for diverse localized application scenarios:
Explore our Titanium Orthopedic Surgical Implant Suprapatellar Approach Tibial Nail for the latest in minimally invasive surgical techniques.
The company places great importance on scientific and technological innovation. Our strategy focuses on "blue ocean differentiation," particularly in limb and pediatric orthopedics. We hold 27 national utility model patents and 6 invention patents.
With over 8,000 product specifications in orthopedic trauma and spinal implants, we provide a "one-stop shop" for surgical instruments and implants, ensuring that complex procurement needs are met with a single, reliable partner.
Every Intramedullary nail undergoes rigorous mechanical testing. Our products, exported to countries like Peru and Morocco, are synonymous with durability and surgical precision, ensuring zero-defect performance in the OR.
The engineering behind our Interlocking Intramedullary Nails involves complex load-sharing principles. Unlike plates that take the full load, an IM nail shares the stress with the cortical bone, promoting the formation of external callus. Our "Expert-Level" designs include multi-planar locking options at the distal and proximal ends, providing unmatched rotational stability.
For proximal femur fractures, our PFNA Femoral Implant features a helical blade that compacts the bone during insertion, rather than removing it. This is crucial for geriatric patients where bone density is a concern. The blue, green, and gold color-coded anodization not only looks high-tech but serves as a vital tool for surgeons to quickly identify sizes and types during high-pressure trauma surgeries.

Our marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. We have built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. We actively engage in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.


As the industry moves toward Smart Implants and biodegradable materials, our factory remains at the forefront of this evolution. By combining our ISO-certified manufacturing processes with a deep understanding of surgeon needs, we continue to serve as a world-class Intramedullary Nails Supplier & Exporter. Whether it is improving the design of a cannulated nail or expanding our reach to new markets in Southeast Asia and Europe, our goal remains constant: providing the tools that help patients walk again.
Our commitment to quality control, combined with a robust production capacity covering more than 8,000 specifications, ensures that we are not just a supplier, but a long-term partner in the global healthcare ecosystem. From the first founded year in 2009 to our current standing as an industry leader with 30+ IP certificates, we invite global agents and hospitals to experience the difference of precision engineering from China.