 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
As a leading Intramedullary Nails Manufacturer & Exporter in Kenya, we recognize the transformative shift in the East African medical landscape. Kenya has emerged as a regional hub for healthcare excellence, with Nairobi hosting some of the most advanced trauma centers in Africa. The demand for high-quality intramedullary (IM) nails has surged significantly due to the rise in modernization, infrastructure development, and unfortunately, an increase in road traffic accidents involving "Bodabodas" and motor vehicles.
Modern orthopaedic surgery in Kenya is evolving. Surgeons in public hospitals like Kenyatta National Hospital and private institutions such as The Aga Khan University Hospital are increasingly moving towards minimally invasive techniques. Our Intramedullary nails provide the stability and biological environment necessary for rapid bone healing, which is crucial for the Kenyan workforce to return to productivity as quickly as possible.
The industry is moving toward "Smart Implants" and anatomically contoured designs. In Kenya, the use of PFNA (Proximal Femoral Nail Antirotation) systems has become the gold standard for trochanteric fractures. These nails offer superior rotational stability, which is vital for elderly patients in Kenya who are prone to osteoporosis-related hip fractures.
Furthermore, the shift from stainless steel to Titanium Alloy (Ti6Al4V) is evident. Titanium offers better biocompatibility and a modulus of elasticity closer to human bone, reducing the "stress shielding" effect. Our exports to Kenya prioritize these advanced materials to ensure long-term patient success and minimize the need for revision surgeries.

Year Founded

Exp. Countries Including Kenya

Serving Top-tier Hospitals

Intellectual Property Rights

Global Agents
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas. Our presence in Kenya is supported by this massive manufacturing capability, ensuring that we can supply any hospital from Kisumu to Mombasa with the exact specifications required for their unique patient cases.
Our marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities globally. We have built a nationwide marketing network covering 31 provinces and cities in our home base, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. We are now expanding this successful model to Kenya, engaging in academic promotion, technical services, and product training for Kenyan orthopaedic doctors.
The company's self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, Morocco, and now expanding rapidly in Kenya. We participate in or host more than 50 academic conferences for orthopedic doctors annually to ensure our technology stays at the cutting edge.
Kenya's unique demographic and geographic challenges require versatile orthopaedic solutions:




Orthopedic PFNA Femoral Implant Nairobi Standard Proximal Femur Interlocking Nail
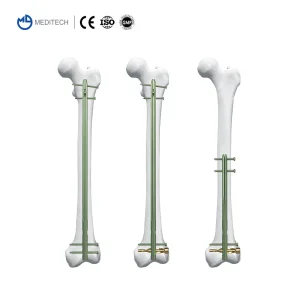
Titanium Orthopedic Suprapatellar Approach Tibial Intramedullary Nail for Kenya

Titanium Cannulated Intramedullary Interlocking Pfna Nails for East Africa

Best Price Expert Tibial Proximal Interlocking Nail System Kenya
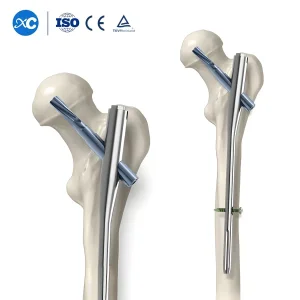
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail Kenya Grade

Intertan Intramedullary Nail for Advanced Fracture Surgery in Nairobi
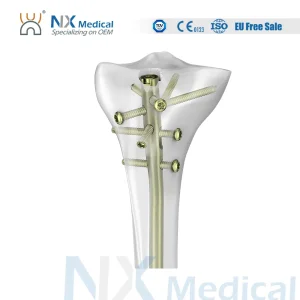
Nx Medical Trauma Proximal Tibial Interlocking Nail - Kenya Factory Direct
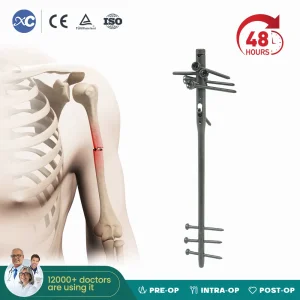
Titanium Multi-Lock Humeral Intramedullary Nail for Kenyan Hospitals
Exporting to Kenya requires a deep understanding of the Pharmacy and Poisons Board (PPB) regulations and the Kenya Bureau of Standards (KEBS) requirements. Our manufacturing facility operates under strict ISO 13485 quality management systems, ensuring every intramedullary nail that reaches Nairobi, Eldoret, or Nakuru is of the highest pedigree.
We utilize advanced CNC machining and surface treatment technologies (such as Type II Anodization for titanium) to ensure our implants provide superior resistance to corrosion and fatigue. For our Kenyan partners, we offer comprehensive surgical kits and instrument sets that are designed for longevity and ease of sterilization in autoclaves typically used in the region.
The future of intramedullary nailing in Kenya is bright. With the expansion of health insurance coverage (NHIF), more Kenyans are gaining access to life-changing orthopaedic surgeries. Our goal is to remain the preferred Intramedullary Nails Manufacturer & Exporter in Kenya, fostering long-term partnerships with distributors, surgeons, and healthcare administrators. We provide not just products, but a complete support ecosystem, including surgical training videos, on-site technical assistance, and a robust supply chain to prevent stock-outs in critical trauma situations.
Contact our Kenya export division today to discuss bulk procurement, agency opportunities, or hospital supply tenders.
Send Inquiry Now