 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
Understanding Market Dynamics and Technological Frontiers
The global maxillofacial implants market is undergoing a transformative era, driven by the increasing incidence of facial trauma, congenital abnormalities, and the rising demand for minimally invasive reconstructive surgeries. As premier maxillofacial implants manufacturers, we observe a significant shift from standardized off-the-shelf components to highly personalized, patient-specific solutions. The integration of 3D printing (additive manufacturing) and biocompatible materials like Medical-Grade Titanium and PEEK (Polyetheretherketone) has redefined surgical outcomes.
In regions such as North America and Europe, the market is characterized by high adoption rates of computer-aided design and manufacturing (CAD/CAM). Meanwhile, the Asia-Pacific region is emerging as a manufacturing powerhouse, with Chinese factories leading the way in balancing high-tech innovation with cost-effective production scales. The global industry is currently valued in the billions, with a projected compound annual growth rate (CAGR) that reflects the aging global population and improved healthcare infrastructure in developing nations.
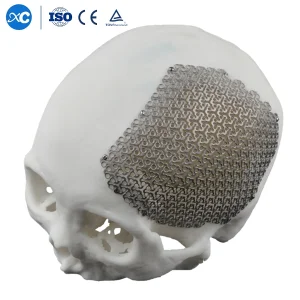
Our 3D Anatomical Cloud Shape CMF Cranial Mesh represents the pinnacle of titanium implant technology. Designed to match the complex contours of the human skull, this implant reduces surgical time and improves aesthetic symmetry for patients undergoing major reconstructive procedures.
Innovating for the Next Generation of Maxillofacial Surgery
The use of AI-driven software to simulate surgeries before the first incision is made. This ensures that the implants manufactured fit perfectly with the patient's unique anatomy.
Research into polymers that provide temporary fixation and then dissolve once the bone has healed, eliminating the need for a second removal surgery.
Greater precision in the placement of micro-plates and screws, reducing tissue damage and accelerating patient recovery times.
What Hospitals and Distributors Look For in a Manufacturer
Procuring medical implants is a rigorous process involving strict regulatory compliance and quality assurance. Global B2B buyers—ranging from large hospital groups in the Middle East to specialized distributors in Latin America—prioritize the following:

Our Locking Titanium Mini Maxillofacial Plates are the workhorses of facial trauma surgery. With low-profile designs to minimize soft tissue irritation and a wide variety of shapes (L, T, Y, and straight), they offer surgeons unparalleled flexibility.
Blending Craftsmanship with Large-Scale Industrial Efficiency
As a specialized manufacturer based in China, we leverage a unique ecosystem that combines advanced R&D with an efficient supply chain. Our facility is equipped with state-of-the-art CNC machining centers and 3D printing labs dedicated to orthopedic and maxillofacial excellence.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.

Year Founded

Exp. Countries

Top-tier Hospitals

IP Rights

Global Agents
Versatile Solutions for Complex Surgical Challenges
Our titanium plates are used extensively in emergency rooms worldwide to stabilize fractures of the zygomatic bone, maxilla, and mandible resulting from traffic accidents or sports injuries.
For patients requiring jaw realignment, our sagittal split plates and LeFort I plates provide the rigid fixation necessary for successful bone healing and functional alignment.
In cases of oral cancer requiring segmentectomy, our custom reconstruction plates (Patient-Specific Implants) allow for the bridging of large bone gaps with high anatomical accuracy.
Small-scale, low-profile plating systems specifically designed for the delicate bone structures of pediatric patients, ensuring minimal growth interference.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
Our self-operated export business is also taking shape, with sales extending to countries such as Brazil, Colombia, Peru, Egypt, and Morocco. We are not just a maxillofacial implants manufacturer; we are a partner in global health, providing the tools necessary for surgeons to restore form and function to their patients.


