


1 / 5
 Alur Orthopedic Implants
Alur Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |
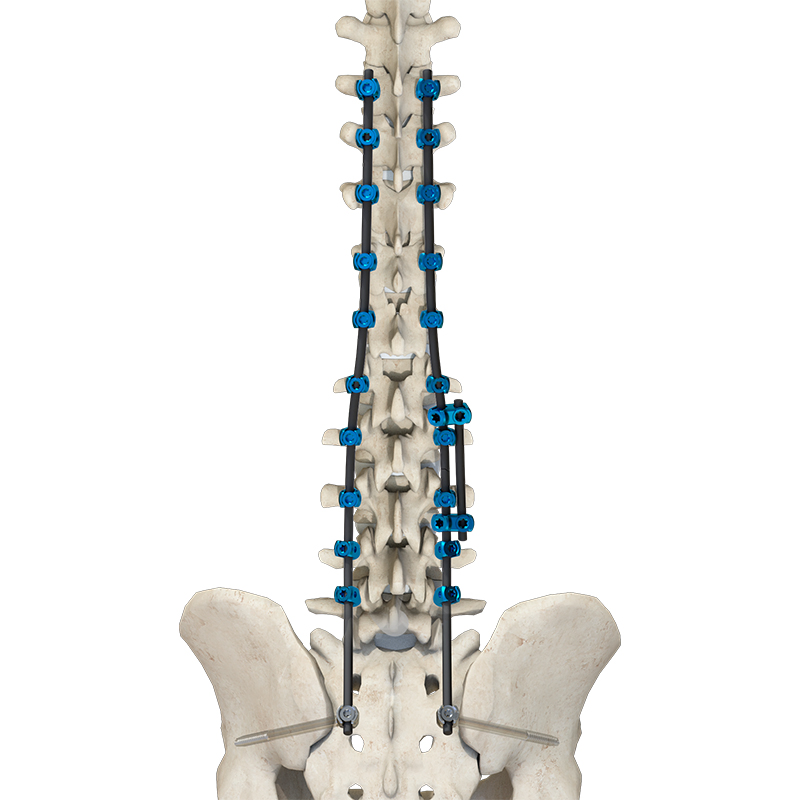
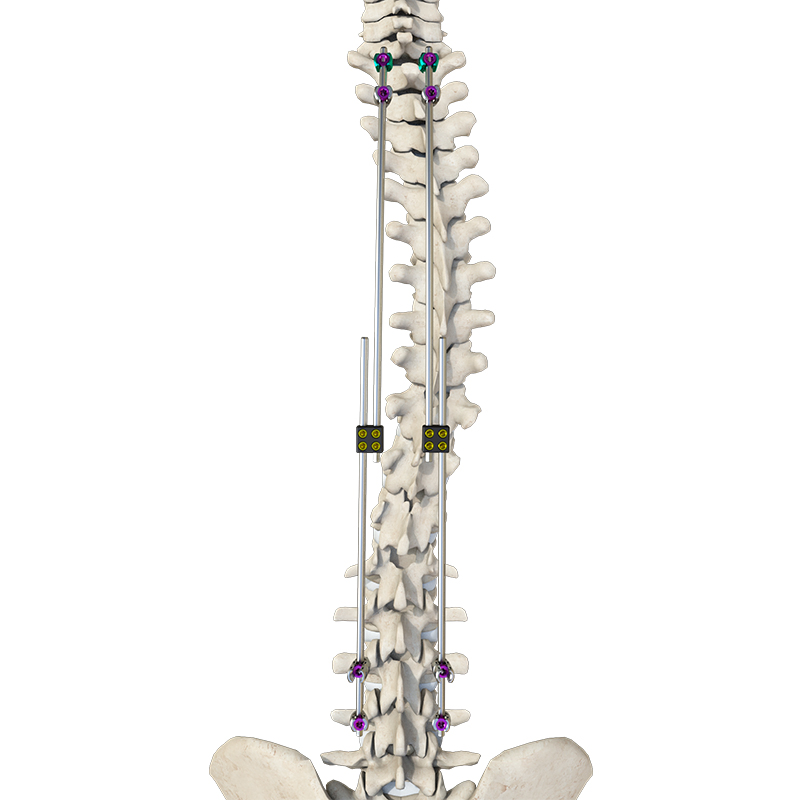
Components are crafted from TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G) for maximum durability and biocompatibility.


| Model Number | Spec. (mm) |
|---|---|
| 1072301 | S |
| M |

A Leading Manufacturer You Can Trust
Our focus is on providing standardized orthopedic implants and instruments for global distributors. We offer a full product range covering trauma, spine, joints, sports medicine, and CMF — all supported by complete surgical instrument sets.
With in-house R&D, certified laboratories, and modern production lines, we support partners who need private label or custom solutions, ensuring reliable and scalable manufacturing.