🔬 Product Description
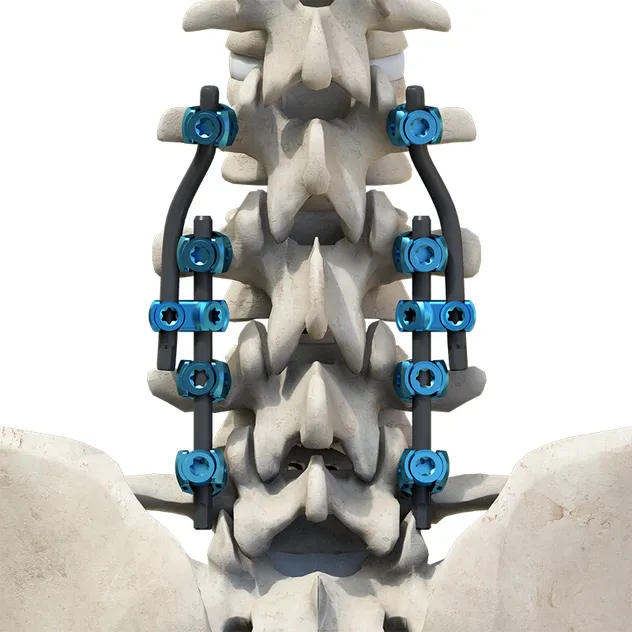
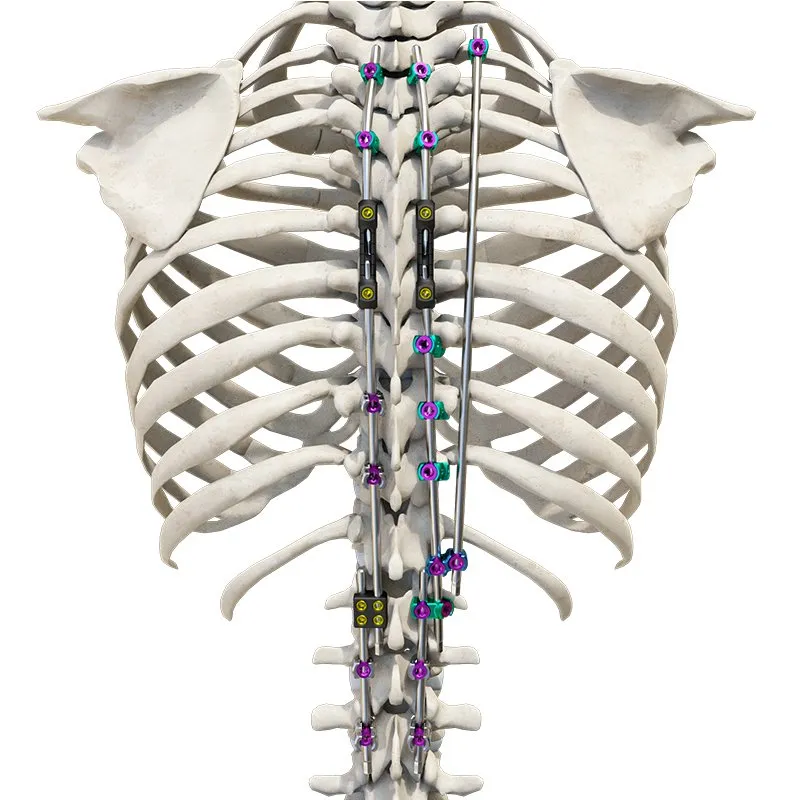
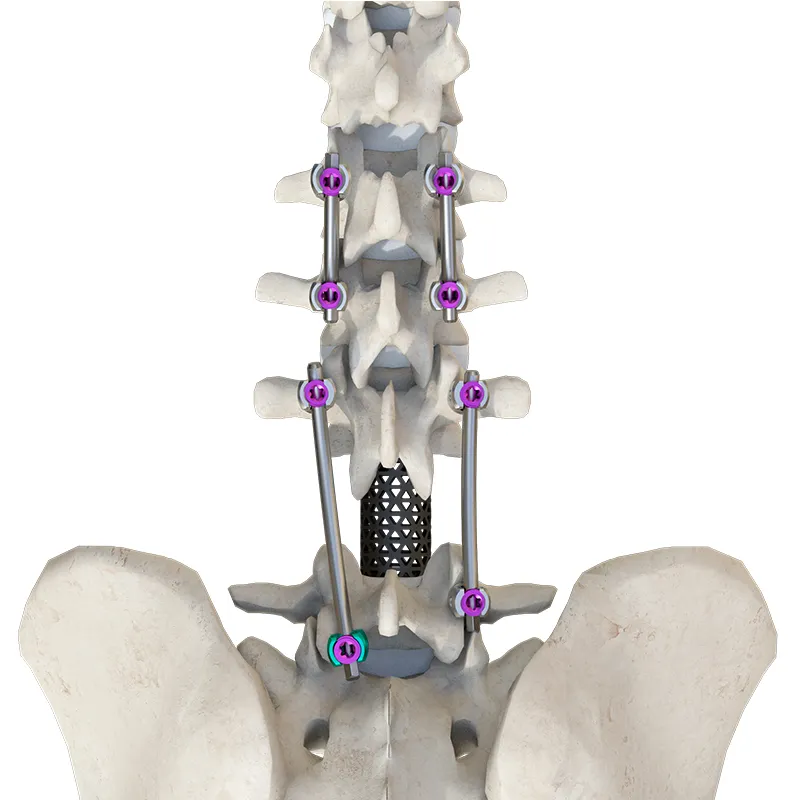
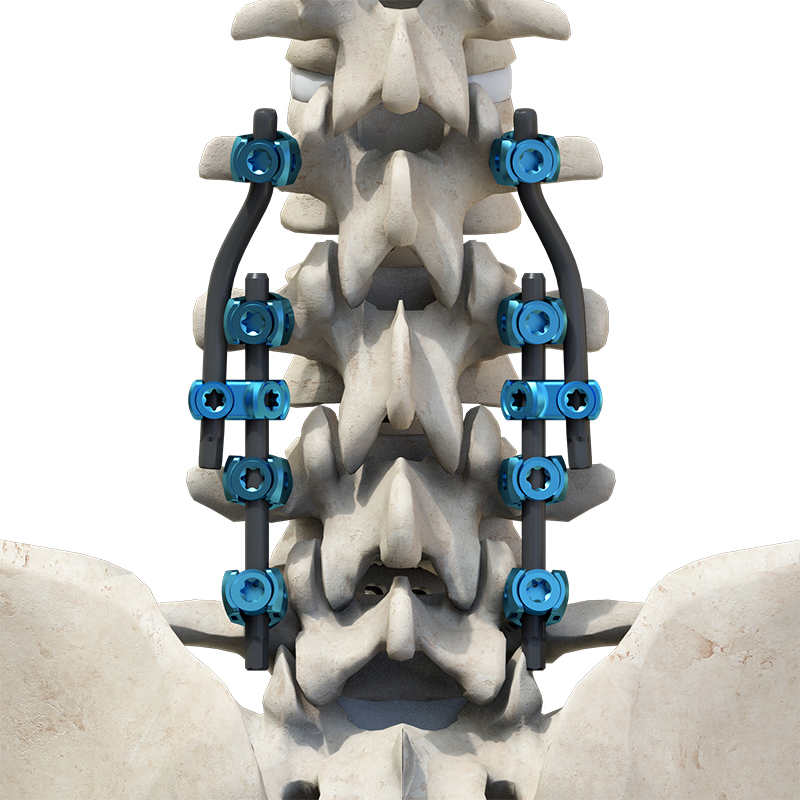
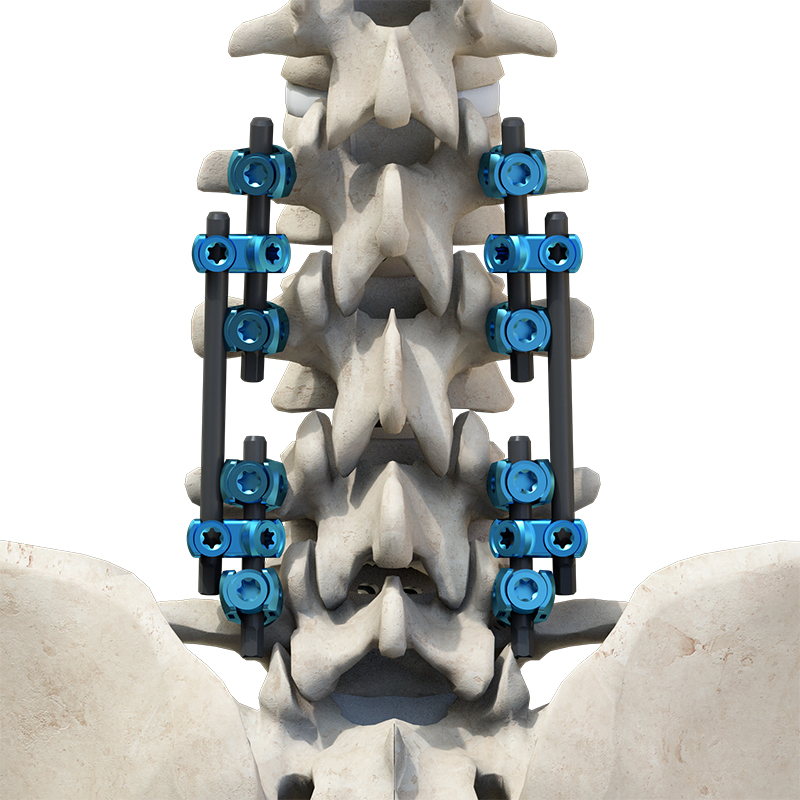
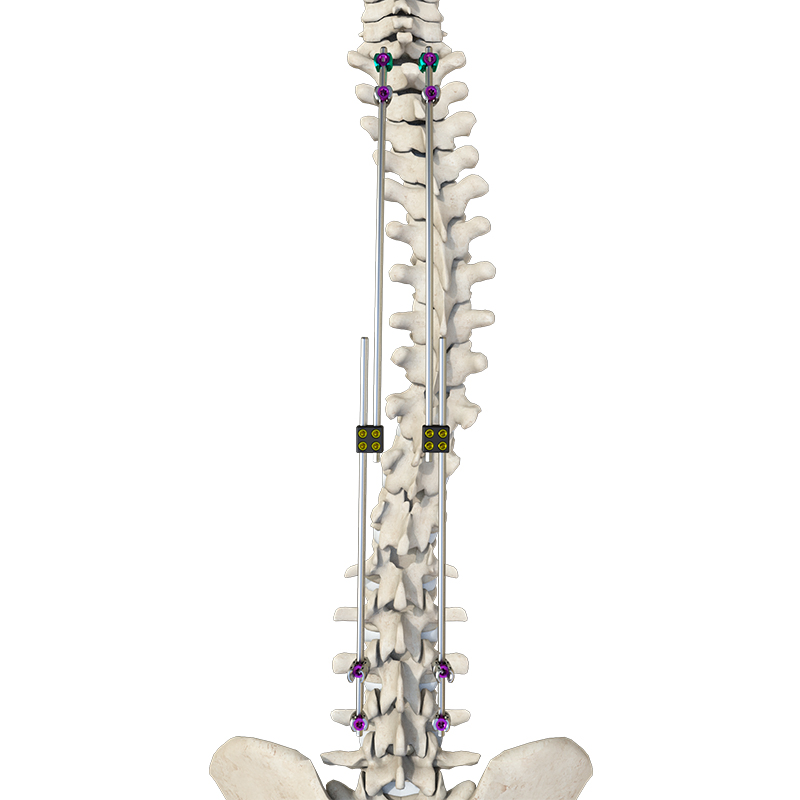
The Thoracolumbar Posterior Pedicle Screw and Rod System (CoCrMo Alloy) consists of pedicle screws, rods, set screws, crosslinks, lateral connectors, hooks, and other accessories.
This system is designed to restore spinal alignment, correct deformities, and provide immediate and long-term biomechanical stability following posterior thoracolumbar procedures. By placing pedicle screws through the vertebral pedicles and connecting them with contoured rods and various components, the system allows for distraction, compression, derotation, and realignment maneuvers.
The assembly forms a stable three-dimensional construct that supports load-sharing and facilitates spinal fusion.
Materials: Components are made of TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G).
✅ Indications
The system is intended for posterior thoracic, lumbar, sacral, and sacroiliac spinal fixation, fusion, and deformity correction in patients with the following conditions:
Vertebral fractures and traumatic spondylolisthesis
Correction of post-traumatic malunion or nonunion
Severe or recurrent lumbar disc herniation
Spinal instability or spondylolisthesis
Post-laminectomy instability due to spinal stenosis
Stabilization after resection of vertebral tumors
Inective for intradural tumors
Post-debridement stabilization in discitis
Reconstruction after spinal tuberculosis debridement
Correction of congenital scoliosis
Surgical treatment of idiopathic scoliosis
Kyphotic deformity correction
🏢 Manufacturing Excellence
We are a leading manufacturer of standardized orthopedic implants and instruments for global distributors. Our professional orthopedic range covers trauma, spine, joints, sports medicine, and CMF.
All products are supported by complete surgical instrument sets, designed for reliability and scalability. With in-house R&D, a certified lab, and modern production lines, we provide high-quality medical solutions that meet stringent global standards.
❓ Frequently Asked Questions
Q: What quality certifications do your products carry?
A: We hold the EN ISO 13485 certification, ensuring our manufacturing processes and medical devices meet the highest international standards for safety and performance.
Q: What are the primary materials used in the Thoracolumbar System?
A: The system utilizes high-grade materials including TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G) for optimal biocompatibility and strength.
Q: Can you accommodate custom or modified product orders?
A: Yes, we accept custom orders. By providing pictures or samples of the required design, our R&D team can work to create similar items tailored to your specific market needs.
Q: What are the typical shipping methods and costs?
A: We primarily use professional courier services for global delivery. Shipping costs are calculated based on the destination, package volume, and weight. Detailed quotes are provided upon request.
Q: Do you maintain ready stock for your catalogue items?
A: Yes, we maintain stock for most standard products with regular monthly production cycles. For specific high-volume requirements, it is best to consult with our sales team for current inventory status.
Q: What is the minimum order requirement for new clients?
A: Our standard minimum order value is US$1000. However, we offer sample orders at cost to allow potential partners to evaluate our product quality before moving to larger volumes.

 Alur Orthopedic Implants
Alur Orthopedic Implants