Q1: What are the main materials used for the screws?
A: The screws are primarily manufactured using high-grade Titanium Alloy and Stainless Steel to ensure excellent biocompatibility and strength.
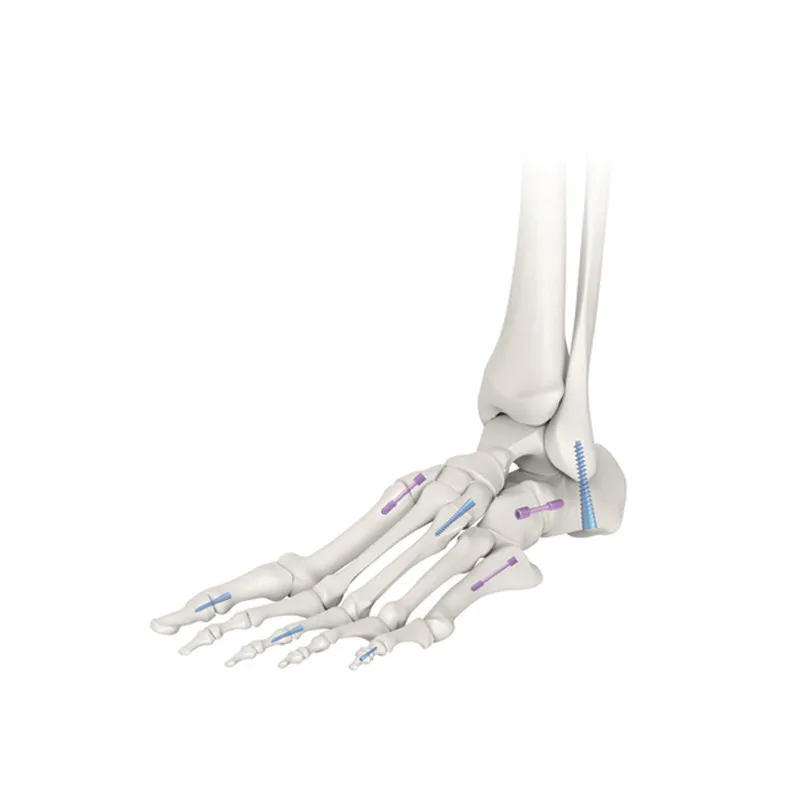
Q2: What clinical applications are these cannulated screws for?
A: They are commonly used for spine fractures and various orthopedic bone fixation procedures.
Q3: Do your products have international quality certifications?
A: Yes, our products are certified by CE and ISO13485, meeting global medical device standards.
Q4: What is your monthly production capacity for these implants?
A: Our current production capacity is approximately 10,000 pieces per month.
Q5: What are the available shipping methods?
A: We offer multiple transportation options including DHL, TNT, EMS, UPS, FedEx, and Air freight.
Q6: Can you provide customized sizes or OEM services?
A: Yes, we provide OEM services and can accommodate specific model/specification requirements as detailed in our product catalog.





 Alur Orthopedic Implants
Alur Orthopedic Implants