


1 / 5
 Alur Orthopedic Implants
Alur Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |


| Product | Size / Length (mm) | Material |
|---|---|---|
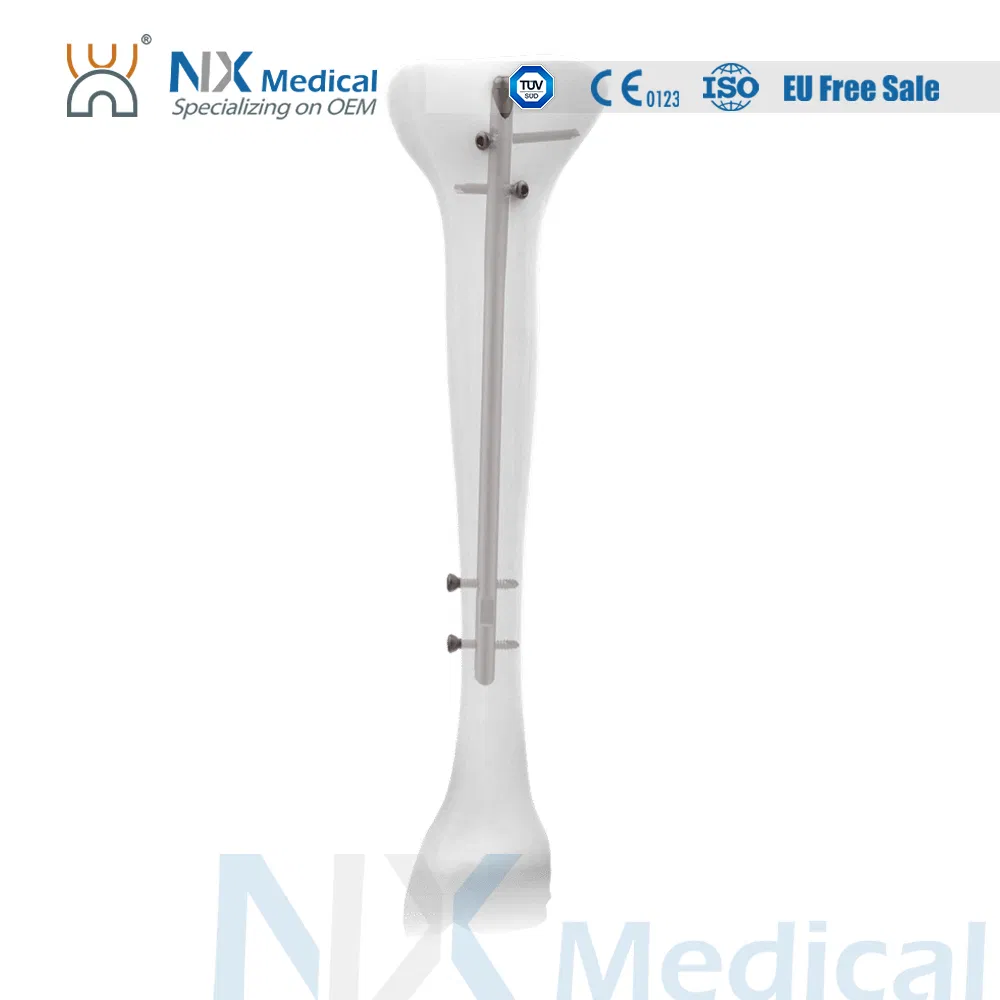
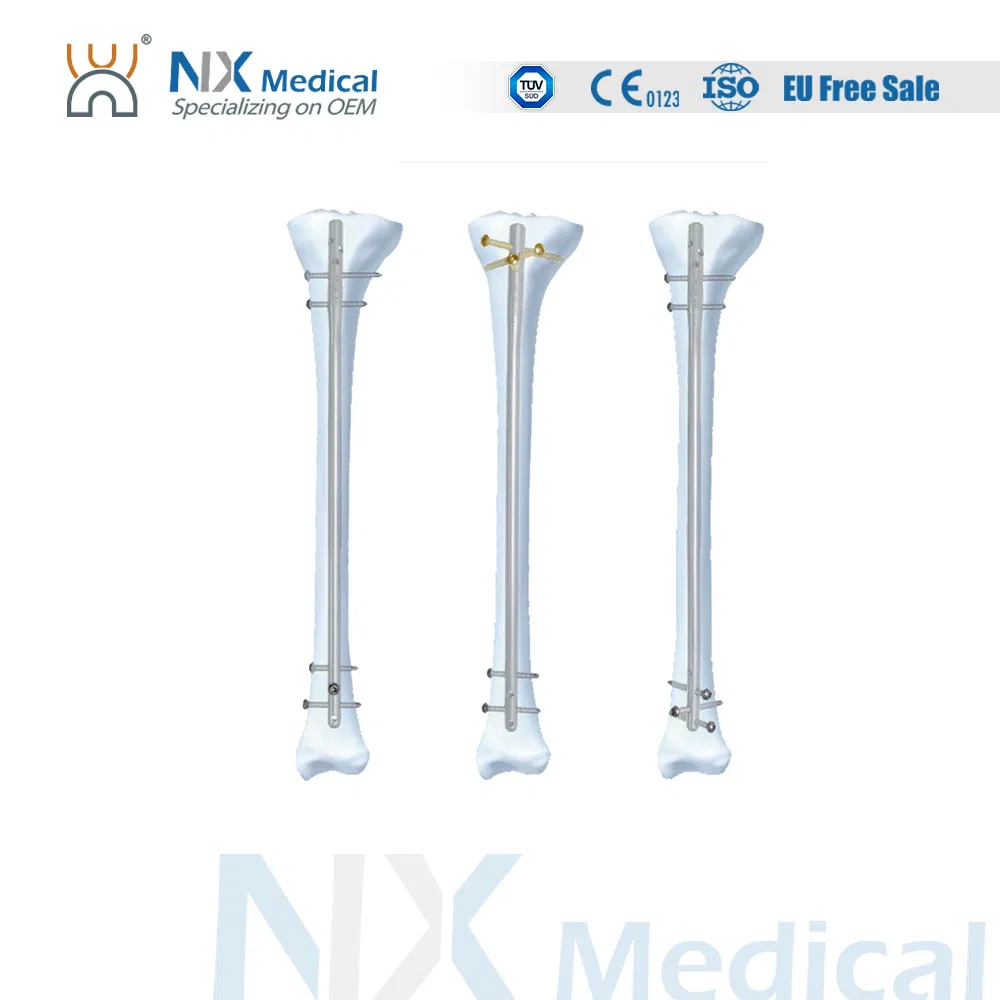
| Tibial Nail | 8 / 9 / 10 / 11 x 255 - 420 (15mm increments) | Titanium Alloy (TA) |
| Cancellous Screw | φ4.8mm | Length: 25 - 90mm | Titanium Alloy (TA) |
| Interlocking Screw | φ4.4mm | Length: 24 - 88mm | Titanium Alloy (TA) |
| Tibial Nail End Cap | Extensions: 0, 5, 10, 15mm | Titanium Alloy (TA) |
A leading Orthopedic product manufacturer located in East China, providing a comprehensive range of cost-effective surgical and reliable therapeutic solutions to surgeons and patients. The facility offers superior various kinds of trauma implants, spinal implants, and surgical instruments worldwide.
The trauma system includes major categories such as locking plates, intramedullary nails, cannulated headless screws, and cable systems. All products are manufactured under a strict quality system to ensure patient safety and clinical efficacy.




