



1 / 5
 Alur Orthopedic Implants
Alur Orthopedic Implants










| Customization: | Available |
|---|---|
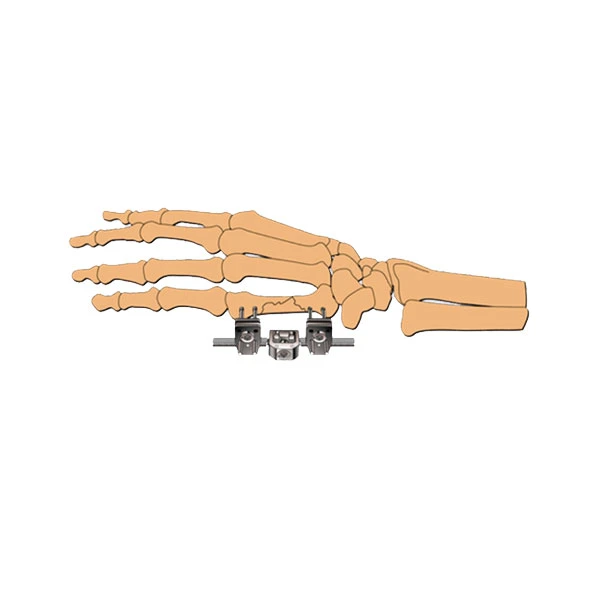
| Type: | External Fixation |
| Application: | Orthopedic |
| Code | Specification | Component |
| D501 | Long 85mm | Fixator Body |
| D502 | Middle 75mm | Fixator Body |
| D503 | Short 65mm | Fixator Body |
| Schanz Pin Details | ||
| Diameter: 2.0mm | Specification (mm) screw length / thread length | |
Material: Aluminum
Surface Finish: Black (Customized options available)
Certificate: CE & ISO




