




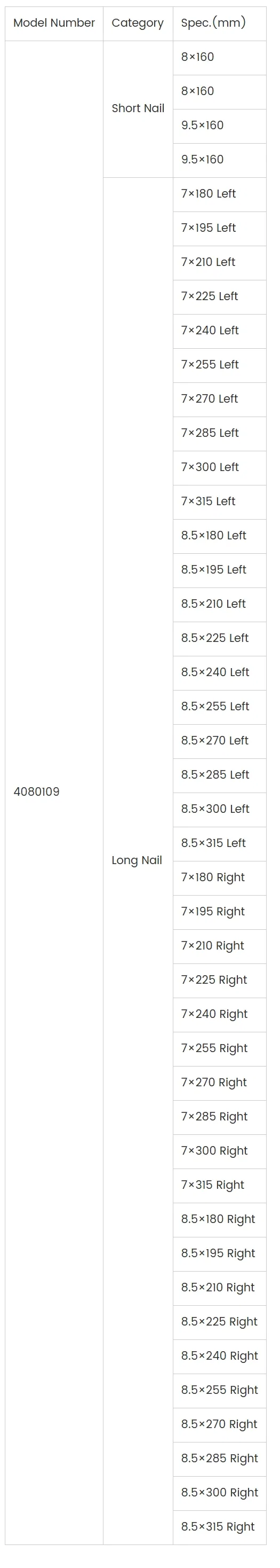
 Alur Orthopedic Implants
Alur Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 108/Piece Request Sample |
Proximal humeral fractures are among the most common fractures, accounting for 4%–5% of all fractures. Mechanism of injury: Usually occurs when the elbow strikes the ground or the arm is braced during a fall, especially in elderly patients, or when the shoulder joint directly sustains external force.

"Screw-in-screw" technique inserted through the head of lateral screws to enhance stability, particularly effective for osteoporosis patients.
Three screws inserted laterally into the greater tuberosity ensure basic structural stability; additional anterior screws enhance the construct.
Ascending screws support the medial calcar region. Two distal interlocking screws are positioned in different planes to reduce implant toggling.
Standardized Orthopedic Implants and Instruments
We are a leading manufacturer of orthopedic solutions covering trauma, spine, joints, sports medicine, and CMF. All products are supported by complete surgical instrument sets, designed for low MOQ and fast turnaround.
Equipped with in-house R&D and certified labs, we focus on reliable, scalable manufacturing to support global distribution and custom OEM/ODM requirements.
