




 Alur Orthopedic Implants
Alur Orthopedic Implants










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
Widely used for different bone fracture of different bones.
Hexagonal head for secure fixation.
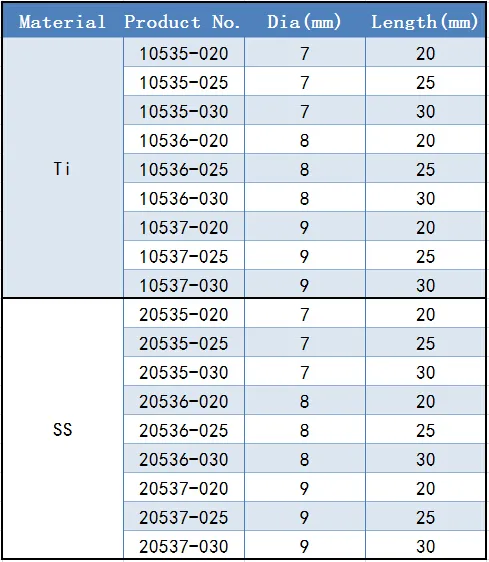
Dia: 7.0mm / 8.0mm / 9.0mm
Length: 20 / 25 / 30mm
As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. This allows for the transformation of clinical experience and innovative ideas into achievements, launching several innovative products with national invention patents and utility model patents.
Revolutionary innovative technologies include the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plate, which are fully endorsed by experts and the market.

Operations are strictly managed according to the ISO13485 quality system, medical device manufacturing quality management specifications, and the CE MDD 93/42/EEC directive.
Advanced processing equipment is utilized, including German-imported and American-imported machining centers, and Japanese-imported automatic lathes, ensuring first-class quality. The dedicated Testing Center features:

Products are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback confirms that product quality consistently meets international standards.