-
✔
Application: Widely used for different bone fractures across various anatomical locations.
-
✔
Available Head: Precision-engineered Hexagonal head for secure fixation.
-
✔
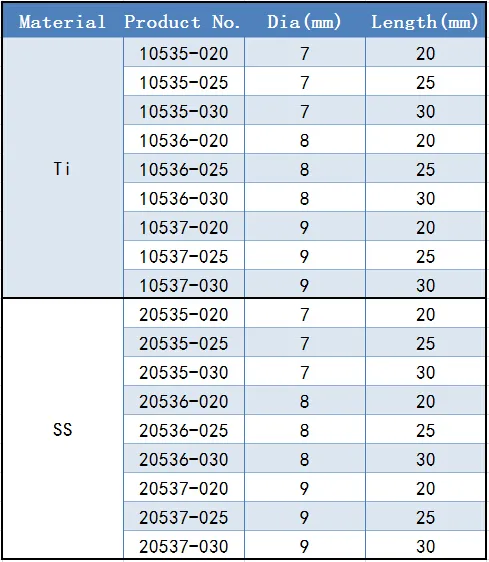
Diameter Options: Available in 7.0mm, 8.0mm, and 9.0mm.
-
✔
Length Variations: Standard lengths include 20mm, 25mm, and 30mm.
-
✔
Instruments: Compatible with specialized ACL Interference Screw Instruments.
🏭 Manufacturing & Technology
As a high-tech enterprise, the investment in R&D exceeds 8% of total sales annually. This commitment facilitates the transformation of clinical expertise into patented innovative medical solutions. Key advancements include the revolutionary Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plate systems, which are recognized as benchmarks in the domestic medical device industry.
Quality Philosophy: "Good faith as the base, quality above all." Operating strictly under ISO13485 quality systems and CE MDD 93/42/EEC directives, every process is standardized to international levels.
🌟 Advanced Equipment & Testing:
Equipped with German and American-imported machining centers and Japanese-imported automatic lathes. The dedicated Testing Center features:
- • Biomechanical Laboratory
- • Physical-Chemical Testing Center
- • Critical Points: Raw material control, product quality control, and performance analysis.
🌎 Global Presence
Our orthopedic solutions are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback from international markets confirms that our products consistently meet and exceed local regulatory standards.





 Alur Orthopedic Implants
Alur Orthopedic Implants