1 / 5
 Alur Orthopedic Implants
Alur Orthopedic Implants










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
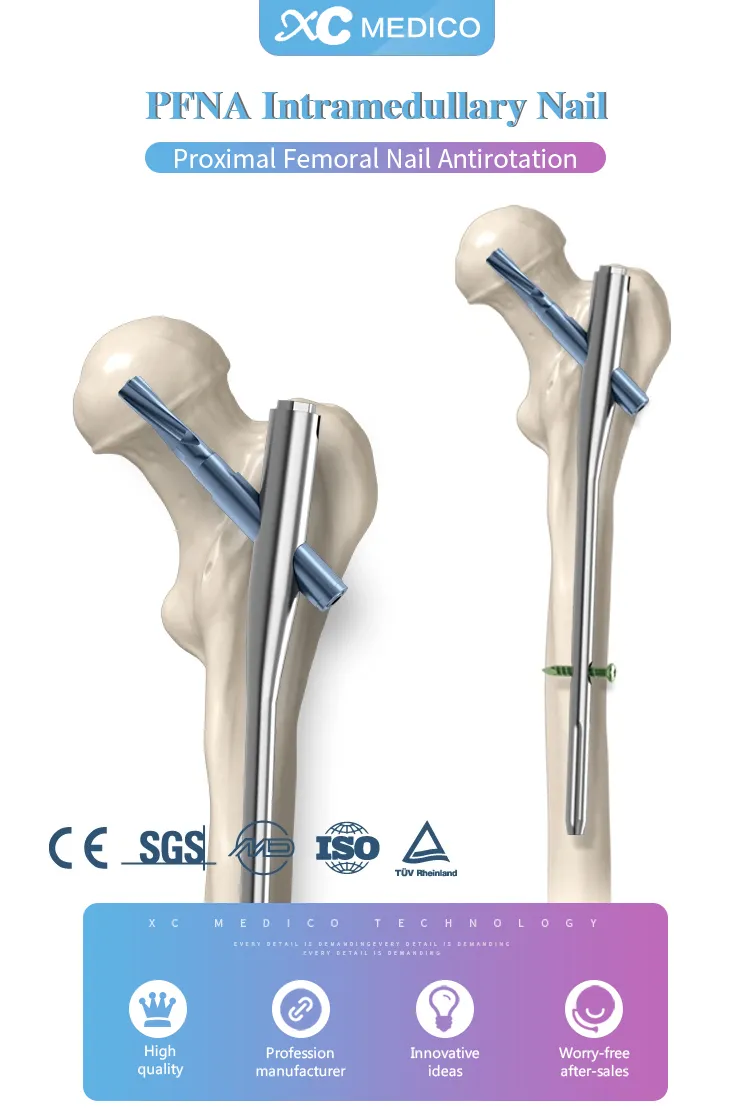


| Product | Specification (Diameter x Length) |
|---|---|
| PFNA(Gama) Intramedullary Nail | Ф9×180 / Ф9×240 |
| Ф10×180 / Ф10×240 | |
| Ф11×180 / Ф11×240 | |
| Ф9×320 to Ф9×420 (R/L) | |
| Ф10×320 to Ф10×420 (R/L) | |
| Ф11×320 to Ф11×420 (R/L) |