




 Alur Orthopedic Implants
Alur Orthopedic Implants










| Customization: | Available |
|---|---|
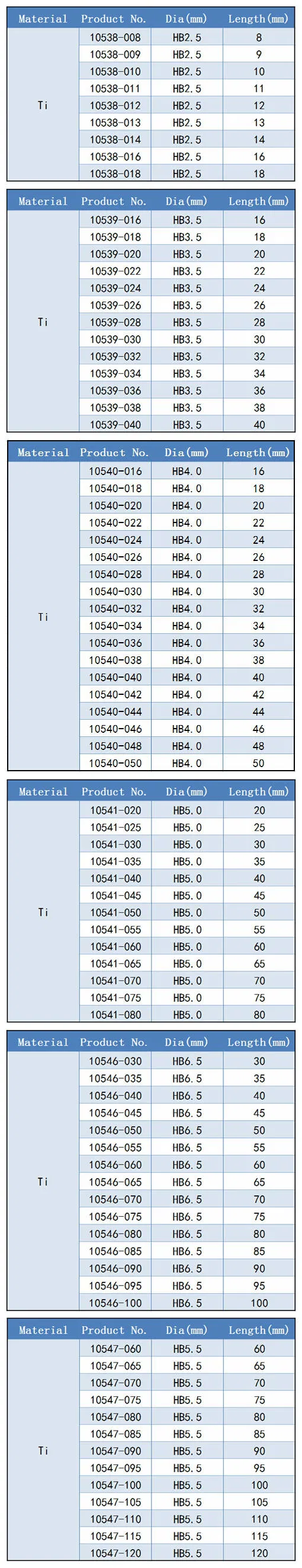
| Type: | Medical Screw |
| Application: | Orthopedic |


As a national high-tech enterprise, R&D investment consistently exceeds 8% of total sales. This focus facilitates the transformation of clinical expertise into innovative medical solutions, backed by numerous national invention and utility model patents.
"Good faith as the base, quality above all"
Operations are strictly governed by the ISO13485 quality system, adhering to medical device manufacturing management specifications, sterile medical device execution rules, and the CE MDD 93/42/EEC directive.
Our production facility features advanced German, American, and Japanese machining centers and automatic lathes. The dedicated Testing Center includes a biomechanical laboratory and a physical-chemical testing center, ensuring rigorous control over:
Our products are successfully exported to global markets including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand, meeting diverse international medical standards.