 Alur Orthopedic Implants
Alur Orthopedic Implants
 Alur Orthopedic Implants
Alur Orthopedic Implants
Driven by innovation, precision manufacturing, and a commitment to improving patient outcomes — delivering world-class orthopedic solutions since 2009.





A trusted orthopedic solutions provider with over 15 years of manufacturing excellence, innovation, and global reach.
The company places great importance on scientific and technological innovation, as well as new product research and development. Its product development strategy follows the principles of "blue ocean, differentiation, and focus strategy," with a strong emphasis on limb orthopedics, pediatric orthopedics, and internal and external fixation products for foot and ankle surgery. The company has obtained 27 national utility model patent certificates, 6 invention patents, and 2 software copyrights.
With a production capacity covering more than 8,000 product specifications in orthopedic trauma, spinal implants, and surgical instruments, the company has gradually established competitive advantages in research and development, production and manufacturing, quality control, marketing, and other key areas.
Pioneering underserved orthopedic market segments with innovative product lines that create new demand.
Delivering uniquely engineered implants and instruments that outperform standard alternatives in clinical settings.
Deep specialization in limb orthopedics, pediatric orthopedics, and foot & ankle fixation for superior clinical outcomes.
A dedicated research team drives ongoing innovation, resulting in 30+ intellectual property certificates and growing.
Our state-of-the-art manufacturing facilities are equipped with advanced CNC machining, precision surface treatment, and clean-room assembly lines — ensuring every product meets international medical device standards.



Over 8,000 product specifications across three major orthopedic categories — engineered for precision, durability, and clinical reliability.
| Product Category | Key Products | Specifications | Focus Area |
|---|---|---|---|
| Orthopedic Trauma | Intramedullary nails, locking plates, screws, external fixators | 3,000+ SKUs | Limb fracture fixation, reconstruction |
| Spinal Implants | Pedicle screws, rods, cages, vertebral body replacements | 2,500+ SKUs | Cervical, thoracic & lumbar spine |
| Foot & Ankle Surgery | Calcaneal plates, metatarsal screws, ankle fusion systems | 1,200+ SKUs | Internal & external fixation |
| Pediatric Orthopedics | Pediatric nails, mini plates, flexible fixation systems | 800+ SKUs | Children's bone development support |
| Surgical Instruments | Retractors, drill guides, targeting devices, instrument sets | 500+ SKUs | Complete surgical procedure support |
We have built systematic competitive advantages across every stage of the orthopedic product lifecycle.
Fully integrated production with CNC precision machining, anodizing, passivation, and clean-room packaging — all under one roof.
In-house engineering team with 6 invention patents and 27 utility model patents, continuously developing next-generation implant solutions.
Multi-stage QC process including dimensional inspection, material traceability, biocompatibility testing, and sterilization validation.
Exporting to 32+ countries across South America, Africa, the Middle East, and Southeast Asia with dedicated international support.
Active participation in 50+ orthopedic academic conferences annually, providing surgeons with hands-on training and technical services.
Our robust intellectual property portfolio and regulatory certifications demonstrate our commitment to innovation, safety, and compliance in global markets.
A professional, well-structured marketing and distribution network ensuring product availability, clinical support, and after-sales service across China and internationally.
The marketing team consists of more than 40 professionals, with sales and technical service personnel present in over 20 major cities. The company has built a nationwide marketing network covering 31 provinces and cities, collaborating with more than 300 agents and serving over 300 top-tier teaching hospitals. It actively engages in academic promotion, technical services, and product training, participating in or hosting more than 50 academic conferences for orthopedic doctors annually.
A dedicated team of 40+ marketing professionals with deep clinical knowledge, providing product education and technical support to surgeons and hospital procurement teams.
Trusted by 300+ top-tier teaching hospitals nationwide, our products are routinely used in high-volume orthopedic trauma, spine, and reconstructive surgery departments.
A robust network of 300+ authorized agents spanning all 31 provinces ensures rapid product delivery, local technical support, and regulatory compliance across every region.
Participating in or hosting 50+ orthopedic academic conferences annually, we support surgeons with evidence-based training, cadaveric workshops, and clinical outcome data.
We offer comprehensive OEM services for global medical device brands — from concept design to finished, sterilized, and labeled product — with full regulatory documentation support.
| OEM Service | Details |
|---|---|
| Custom Design & Engineering | Collaborative product development with your engineering team; CAD/CAM design, FEA analysis, and prototype fabrication within 4–8 weeks. |
| Material Selection | Medical-grade titanium alloy (Ti-6Al-4V), 316L stainless steel, PEEK, and cobalt-chrome — all with full material traceability certificates. |
| Surface Treatment | Anodizing, passivation, sandblasting, HA coating, and PVD coating available to meet specific osseointegration and corrosion resistance requirements. |
| Packaging & Labeling | Custom branded packaging, sterile blister packs, IFU documents, and UDI labeling compliant with EU MDR, FDA, and other regional regulations. |
| Regulatory Support | Full technical file preparation, biocompatibility reports (ISO 10993), sterilization validation, and assistance with CE, FDA 510(k), and local registrations. |
| Minimum Order Quantity | Flexible MOQ starting from 50 units per SKU for custom OEM orders; bulk pricing available for orders exceeding 500 units. |
The company's self-operated export business is rapidly expanding, with sales extending to countries across South America, Africa, the Middle East, and Southeast Asia.
Our international team provides dedicated pre-sales consultation, regulatory registration support, and post-sales technical training — ensuring our global partners receive the same high standard of service as our domestic clients.
Our products have been successfully applied in thousands of clinical procedures across leading hospitals — here are a few representative examples of our collaborative work.
A Level-I trauma center utilized our locking plate system for complex tibial plateau fractures in 120+ cases, reporting 98.3% implant integrity at 12-month follow-up and significantly reduced surgical time.
Partnering with a top-tier spine center, our pedicle screw-rod system was used in 80+ multi-level lumbar fusion procedures, achieving excellent radiographic fusion rates and high patient satisfaction scores.
Our anatomical calcaneal plate was adopted by a regional orthopedic hospital for Sanders Type III fractures, demonstrating superior anatomical reduction and low complication rates in a series of 60 cases.
A children's hospital employed our titanium elastic nail system for pediatric femoral shaft fractures in patients aged 4–14, with 100% union rate and no implant-related complications reported in 45 consecutive cases.
Feedback from orthopedic surgeons, hospital procurement directors, and international distributors who rely on our products every day.
"The locking plate system demonstrates exceptional dimensional consistency and surface finish. Our surgical team has been impressed by the precision of the instruments and the reliability of every implant batch."
"We have been distributing their spinal implant range for three years. The product quality, regulatory documentation, and after-sales support are all outstanding. Our hospital clients consistently reorder."
"Their OEM capability is genuinely impressive. From prototype to final CE-marked product in under 6 months — with full technical file support. We've since launched two product lines under our own brand."
"The pediatric flexible nail system is well-designed, easy to implant, and the kids recover beautifully. I've recommended this product to colleagues at three other children's hospitals."
Whether you are a hospital procurement team, an international distributor, or a medical device brand seeking OEM manufacturing — we are ready to support your goals with proven expertise and reliable supply.
Contact Us Today


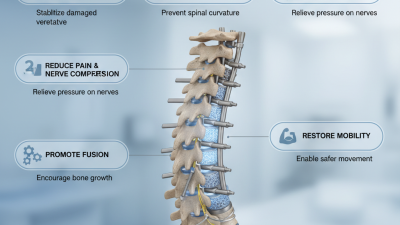
Spinal stability is crucial for overall health and mobility. Many patients wonder, "How do implants improve spinal stability?" Understanding the role
Author: Sienna Date: 2026-03-30
Learn More
The 2026 Canton Fair is set to be a pivotal event for the medical equipment industry, particularly for Hip Replacement Instrument Sets. This
Author: Aria Date: 2026-03-27
Learn More
The upcoming 139th Canton Fair promises to be a significant platform for showcasing innovations in medical equipment, specifically Humeral
Author: Aria Date: 2026-03-25
Learn More